When it comes to developing a commercial vaccine, key factors such as safety and effectiveness play a crucial role, but production costs and ease of use are equally important to determine the product’s success in the market.
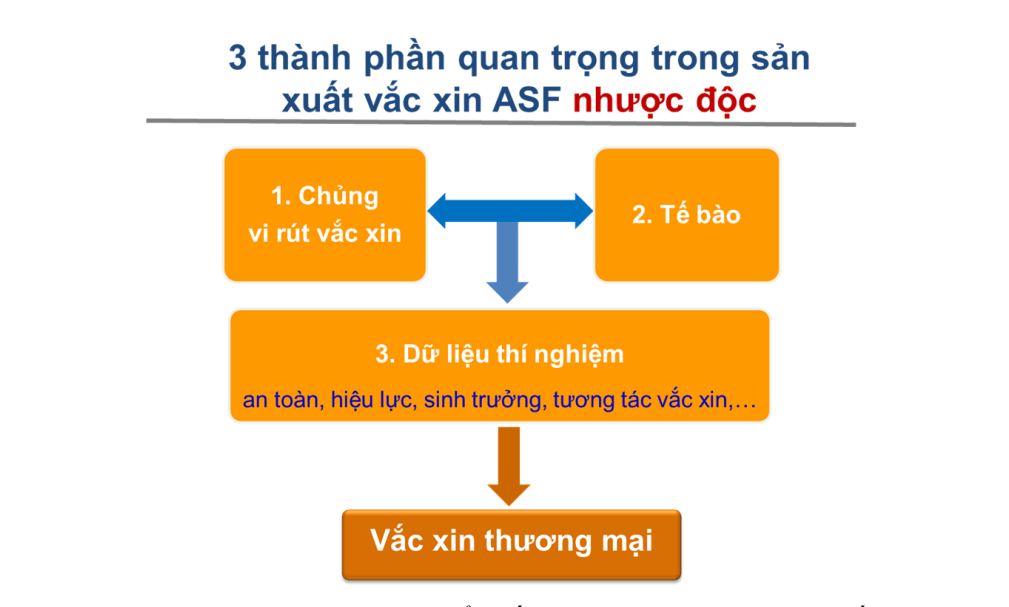
Among the seven technology platforms explored for the development of ASF vaccines, Avac, a Vietnamese vaccine research and development organization, identified a live attenuated vaccine produced through gene editing as the most promising approach. Nguyen Van Diep, the Director General, emphasized that in order to speed up the development of this attenuated vaccine, it is crucial to address three key technical challenges simultaneously: (1) selecting and optimizing a highly safe attenuated virus strain; (2) creating a continuous cell line that enables the stable replication of the ASF virus (ASFV); and (3) establishing a comprehensive database detailing the vaccine’s biological properties, immunogenicity, and safety—this foundational scientific knowledge is vital for moving towards large-scale commercial production. Throughout the project, Dr Diep and his research team progressively overcame these challenges.
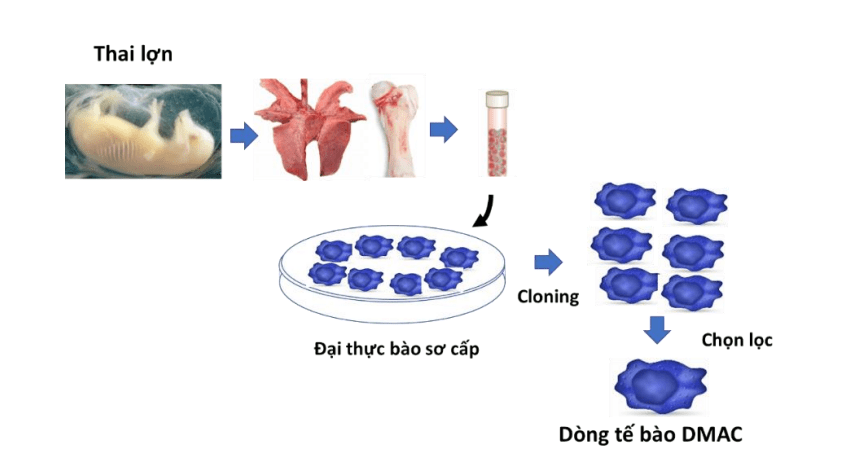
Selection of vaccine virus strains
The three attenuated virus strains developed through the gene deletion method and provided by the United States were subjected to a comprehensive re-evaluation of their biological characteristics. Following this assessment, one virus strain was selected for further optimization. The ASF-G-ΔMGF strain, characterized by the deletion of six virulence genes that belong to two gene families MGF-360 and MGF-505, was continuously cultured on DMAC cells. After 30 generations of culture, selection, and cloning, the research team has established a parent virus strain that meets the stringent criteria of sterility, purity, safety, and high efficacy against the genotype II field virus strain predominantly circulating in Vietnam.
Whole genome pseudo-sequencing results of the vaccine virus strain, which have been submitted to the World Gene Bank under accession number PP529961, showed that the Avac master virus strain exhibits several genetic differences compared to the original strain received from the United States. These differences have led to alterations in the sequence structure of ten proteins, thereby elucidating the mechanisms underlying the attenuation and increased safety of the virus in comparison to the original candidate. From this master virus strain, Avac is generating working seeds to produce antigens for commercial vaccine production batches.
The cell line ensuring ASF virus stability
Dr Diep has successfully developed a novel cell line derived from natural macrophages isolated from porcine embryos. This cell line exhibits continuous growth capabilities, stability in biological properties, and is devoid of oncogenes, which are typically employed to achieve cellular immortality. Furthermore, it is free from contamination by foreign microorganisms and possesses the capability for long-term storage at low temperatures. Notably, this cell line enables the rapid replication of the ASF vaccine virus, attaining high viral titers while maintaining stability in biological characteristics. It serves as a fundamental component in the quality control processes for each vaccine batch produced and is well-suited for the large-scale proliferation demands of industrial production. This cell line has been designated as DMac, an acronym for Diep’s Macrophage, in honor of the inventor, and has been registered for patent protection with the National Office of Intellectual Property.
Comprehensive data on commercial vaccine properties
To ensure safe and effective guidance for farmers and managers, it’s crucial to gather comprehensive information on various aspects of a commercial vaccine product. Avac must continually assess several key factors, including the vaccine’s stability and efficacy, the duration of immunity, interactions with other vaccines, transplacental transmission, and the impact of maternal immunity. Additionally, vaccination protocols should be tailored according to pig age.
Factors like the influence on productivity under varying health conditions in pigs, viral excretion, and horizontal transmission in real farming environments, as well as the presence of virus residues in carcasses and organs, play important roles in evaluating vaccines. It’s also essential to distinguish between vaccine virus strains and field virus strains. To obtain sufficient data, Avac needs to carry out extensive laboratory and animal testing at farming facilities. Each test must be meticulously designed and aligned with the current standards for live vaccines generally, and for ASF vaccines specifically.
Given that there are currently no international standards for ASF vaccines, Avac must establish its own guidelines while also incorporating the highest standards outlined in the latest draft developed by the World Organization for Animal Health (WOAH). After successfully completing over 100 animal tests, which yielded positive results, the company has compiled a foundational dataset on many product criteria that allows for confident recommendations for each specific application.
The results

The research findings indicate that the vaccine strain, consisting of a minimum protective dose of 103 attenuated ASFG-ΔMGF viruses, demonstrates safety for swine aged 4 weeks and older. This vaccine facilitates the rapid development of protective immunity within a timeframe of just 2 weeks, achieving peak protection by 4 weeks post-vaccination. The efficacy rate exceeds 90% among vaccinated pigs, with immunity sustained for over 5 months. Importantly, the vaccine does not adversely affect the pigs’ growth or their immune responses to other vaccinations. Preliminary investigations further suggest that the vaccine is safe and effective for sows and boars.

“In light of these promising results, Avac has submitted an application for approval to commercialize a vaccine specifically designed to prevent ASF in pigs, branded as Avac ASF Live. This vaccine is recommended for swine aged 4 weeks and older. A single injection establishes protective immunity that endures for up to 5 months, effectively encompassing the entire life cycle of commercial pigs. The convenience of a one-time vaccination procedure for post-weaning pigs positions Avac ASF Live as a valuable option for enhancing swine health management.”
Vaccine registration and commercialization
The Avac ASF Live vaccine has passed a comprehensive testing process before being approved for circulation. The Department of Animal Health (DAH) has evaluated the strain and tested three batches of the vaccine to confirm its safety and effectiveness on two farms under strict protocols. Following this, they assessed the vaccine’s safety and protective efficacy on four farms in Bac Giang and Hanoi, where pig populations ranged from 500 to 20,000. The findings showed that the Avac ASF Live vaccine is safe for pigs of all ages (between 4 and 13 weeks), even in instances of overdose (up to 10 doses). Additionally, the vaccine provided an impressive average protection rate of over 80% when confronted with a genotype II field virus strain collected in Vietnam.
On July 8, 2022, the Avac ASF Live vaccine received a license for controlled circulation in Vietnam. After working closely with relevant authorities to evaluate the vaccine’s safety and effectiveness across more than 600,000 doses at nearly 600 farms, it was approved for widespread circulation and export on July 24, 2023. This marks the first ASF vaccine to be authorized by the government for nationwide commercialization and export.

Following the complete commercialization allowance, Avac has continued to coordinate additional trials across more than 20 provinces and cities, involving farms at a scale of 100 to 3000 pigs. The results of these trials have demonstrated that the vaccine is safe and effective.
As of early 2025, Avac has distributed approximately 2.5 million doses of the vaccine to the domestic market, with over 600,000 doses allocated specifically to the Animal Husbandry and DAH and Agricultural Service Centers across various provinces.
In addition to its domestic applications, the Avac ASF Live vaccine has been introduced at numerous prestigious international scientific forums organized by global institutions. These include the Global Alliance for African Swine Fever (GARA), the Food and Agriculture Organization of the United Nations (FAO), the World Organization for Animal Health (WOAH), the International Livestock Research Institute (ILRI), the Philippine Swine Disease Association, the United Soybean Society (USSEC), and numerous academic institutions.
In February 2023, the Bureau of Animal Health (BAI) in the Philippines initiated laboratory trials and conducted tests at ten farms of the Avac ASF Live vaccine, while also assessing its efficacy in Vietnam. The preliminary results indicate that the vaccine is safe and effectively induces strong immunity in pigs. As a result, the Philippine government has authorized the Ministry of Agriculture to import and utilize the vaccine, making the Philippines the first country outside Vietnam to officially approve its use, with 460,000 doses scheduled for import in July 2024. Concurrently, Nigeria has imported 5000 doses for testing and registration. Evaluations and registration processes are ongoing in several other countries, including Nepal, India, Malaysia, Myanmar, Cambodia, Tanzania, Ukraine, Serbia, and Sri Lanka.
Further challenges
During the circulation of a pathogen, it continually alters its biological characteristics, evolves its virulence, and modifies its antigenic properties, which impact vaccine efficacy. Consequently, each vaccine has a distinct life cycle influenced by disease epidemiology and the technologies used in development. The ASFV, in particular, is mutating rapidly, with multiple variants reported in Vietnam.
Since 2021, a recombinant strain formed from genotypes I and II was first identified in China and has since spread to Vietnam, Mongolia, and Russia. Alarmingly, existing commercial vaccines in Vietnam and those under development in China are ineffective against this variant. This evolving challenge necessitates vaccines with broader protection that include this specific strain.
In response, Avac has collaborated with research institutions and is utilizing advanced technologies to swiftly develop new vaccine candidates for the next generation of ASF vaccines.
The vaccination of breeding pigs, including sows and boars, is essential to enhance protection and ensure herd immunity within the pig population. Acknowledging this necessity, Avac has meticulously assessed and refined its ASF vaccine, ensuring its safe and effective use across all pig categories. In 2024, the company partnered with the Central Veterinary Drug Testing Center I and the DAH to develop standards for evaluating ASF vaccines for breeding pigs. This initiative will facilitate the continued evaluation and official licensing of the Avac ASF Live vaccine for breeding pigs in Vietnam in the near future.
Excerpt from an article written by Dr Nguyen Van Diep, General Director of Avac Vietnam JSC.
Translated by Ha Thu

Leave a comment